Comparing the Model S to a low performance, low range Leaf or a low performance also boring Volt is not valid IMHO
I think I can relate to what you mean. That said, this is not about driving fast in a sleek car, which hugs the road. This is about battery technology. I did not hesitate to defend Tesla against what appeared to be baseless sensationalism after the first incident in Kent, WA. Unfortunately, the numbers are starting to reveal that Tesla's battery technology might be at significantly greater risk of thermal events when compared to what the LEAF or the Volt are using.
There is only one Volt fire, which has been reported. In a vehicle that was crash tested and left stored against manufacturer recommendations for three weeks before the fire started. No fires have been reported for the LEAF anywhere. Between these two vehicles, they shipped probably ten times the volume of the Model S and have about ten times more fleet miles with no fire incident. There have been severe accidents involving LEAFs and Volts. One of the ActiveE drivers was involved in a very serious accident, which left the front battery deformed and with a gaping 8-inch hole from hitting a metallic object. The car did not catch fire. BMW uses a chemistry, which is similar to the Volt.
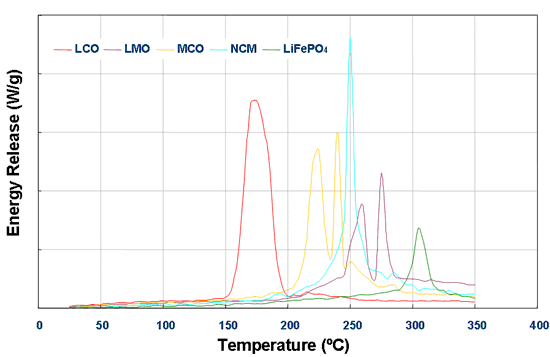
On the following graph the energy release from batteries with LiMn2O4 cathode is shown to be nearly as good as LiFePO4. Hope this helps. I unfortunately don't have much more time to entertain this discussion, but I hope that Tesla will find a way to defuse the situation and identify an appropriate remedy.
 Click to open
Click to open
If you were looking for more authoritative sources of information, Kim suggested the Chemical & Engineering News in his MNL post. A short search yielded the following article. It's worth noting that the LEAF uses a LiMn2O4 cathode and the Roadster used a LiCoO2 cathode. The Model S uses a LiNiCoAlO2 cathode.
Assessing The Safety Of Lithium-Ion Batteries | February 11, 2013 Issue - Vol. 91 Issue 6 | Chemical & Engineering News
Choosing a safe cathode is one key aspect of battery construction. LiCoO2 cathodes developed in the early 1990s made Li-ion batteries the commercial success they are today. Yet it is less stable than other cathode materials. At elevated temperatures, LiCoO2 liberates oxygen, which can react with organic cell components.
LiMn2O4 tolerates heat better than LiCoO2, but the manganese-based material's charge capacity is lower, and it too decomposes at high temperature. In contrast, LiFePO4 stands up especially well to thermal abuse due to the strength of phosphorus-oxygen bonds.


