AlanSubie4Life
Efficiency Obsessed Member
I’d double check and be sure that “new” means “new.”Denise was told she received the new vaccine last week.
For example, Moderna’s 1273.815 vaccine hasn’t had VRBPAC approval and CDC approval and I think it will need both before it can be administered, won’t it?

Moderna (MRNA) Updated COVID Jab Effective Against Eris Variant
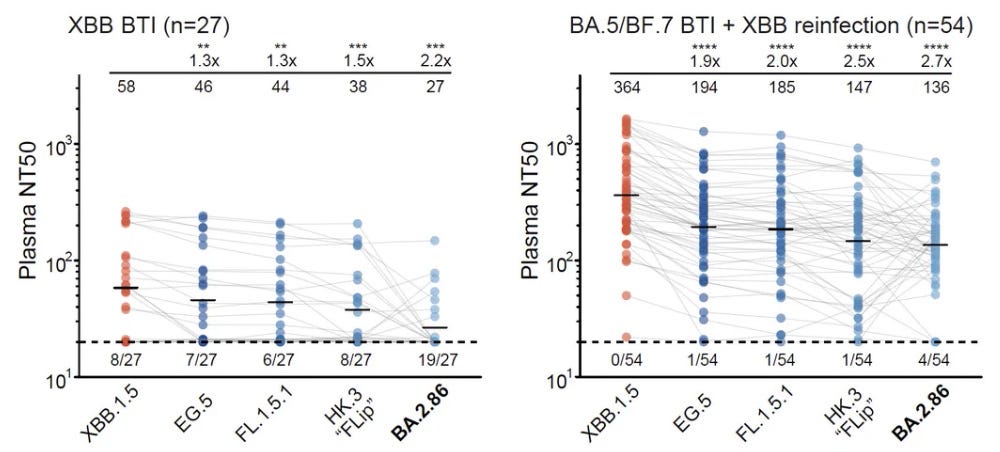
Moderna's (MRNA) updated monovalent COVID-19 vaccine generates a robust immune response against the dominating Eris variant and another rapidly spreading virus strain in a clinical study.
Not sure getting the “old” vaccine matters that much - it’s actually very hard to say, since they do not appear to attempt to check the vaccines for efficacy any more (pretty annoying), nor have any of the bivalent (soon to be monovalent) boosters showed efficacy against infection in any tests that have been done. (Personally I think the reason for this is that it is hard to show efficacy for a booster in the current immune landscape (most have robust protection against infection), but hard to say...) That being said, I am waiting for .815.
Moderna Clinical Trial Data Confirm Its Updated COVID-19 Vaccine Generates Robust Immune Response in Humans Against Widely Circulating Variants
Updated COVID-19 vaccine effectively targets EG.5, a dominant variant of concern, as well as the rapidly spreading FL 1.5.1 variant Updated vaccine expected to be available, pending approval, in coming weeks for fall vaccination season CAMBRIDGE, MA / ACCESSWIRE / August 17, 2023 / Moderna, Inc...
Last edited: